



1 / 5










| Customization: | Available |
|---|---|
| Application: | Biotechnology Industry, Chemical industry, Clinical Diagnosis, Food & Beverage Factory, Food Safety, Forensic Science, Pharmaceutical Industry |
| Certification: | CE, FDA, GMP, ISO |

Leading the Future of Water Treatment Equipment
With a legacy dating back to 1958, our expertise in the core industry of pharmaceutical health spans over 67 years. We remain at the forefront of the pharmaceutical water equipment industry, providing leading solutions and services globally.


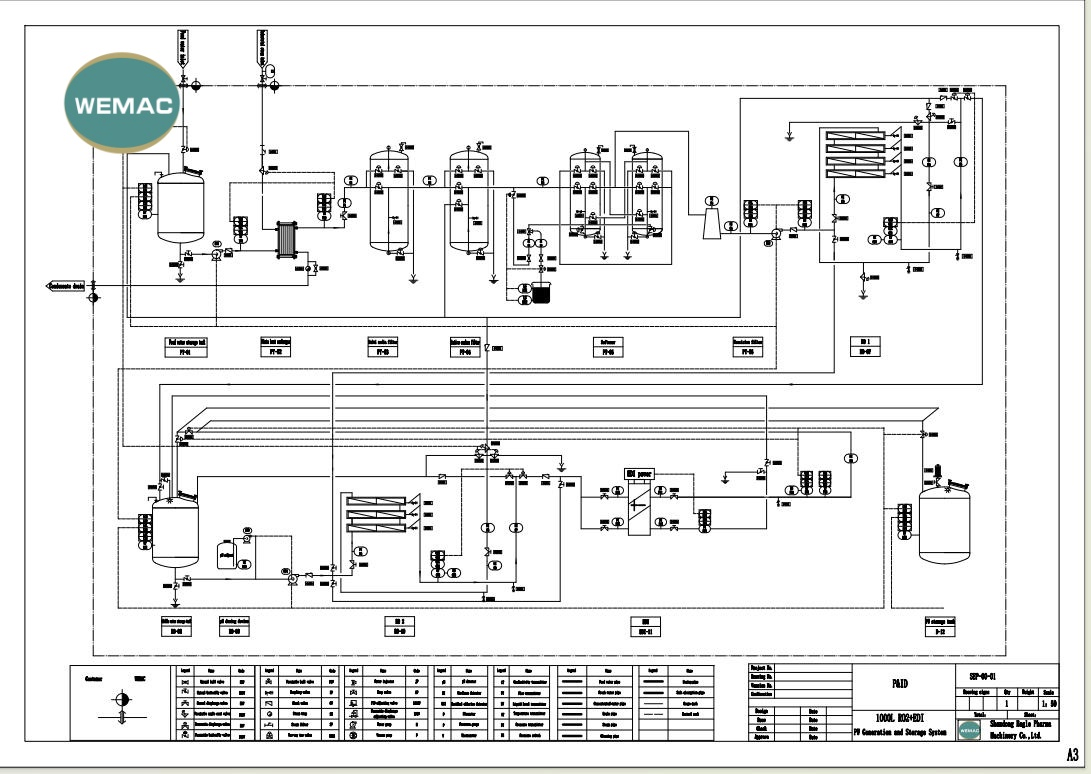
| Design Basis: Water Quality / User Requirement | |
|---|---|
| TDS (Raw water conductivity) | Residual chlorine |
| Hardness | Metal ions (iron, manganese) |
| Microorganism | Turbidity (or SDI) |
| Free CO2 | Nitrite |
| Operating Scenario | Pipeline Flow Direction |
|---|---|
| When produced water is qualified | Produced water enters the purified water storage tank through two pipelines. |
| When produced water is unqualified | Produced water circulates through pipelines back to the middle storage tank. |
| When storage tank is full | System automatically switches to circulation to ensure no dead water remains. |



Our professional process piping design team undertakes clean engineering projects in the biopharmaceutical industry in accordance with GPM specifications, EU standards, and FDA guidelines.





 Hanora Medical
Hanora Medical